In October MCIA released data obtained from the Office of Drug Control that revealed the volume of medicinal cannabis produced by Australian cultivators, along with volumes of imports and exports. MCIA has now obtained data from TGA that sheds light on the nature of products sold in Australia.
The data shows that 696,402 units of medicinal cannabis products were sold in Australia during the first half of 2022. Peter Crock, MCIA Chair said that “This is the first time that data on the number of units of medicinal cannabis products sold in Australia has been made available. On the basis of this data, MCIA estimates that industry revenue for H1 2022 exceeded $100 million.”
Dried flower and inhalable products comprised 73% of all units sold (508,125 units), and oral liquids 26% (181,239 units), with the remainder (1%, 7,038 units) being comprised of capsules, pills, and other products. [FIGURE 1]. The most common size for a unit of dried flower is 10g, retailing at around $140.
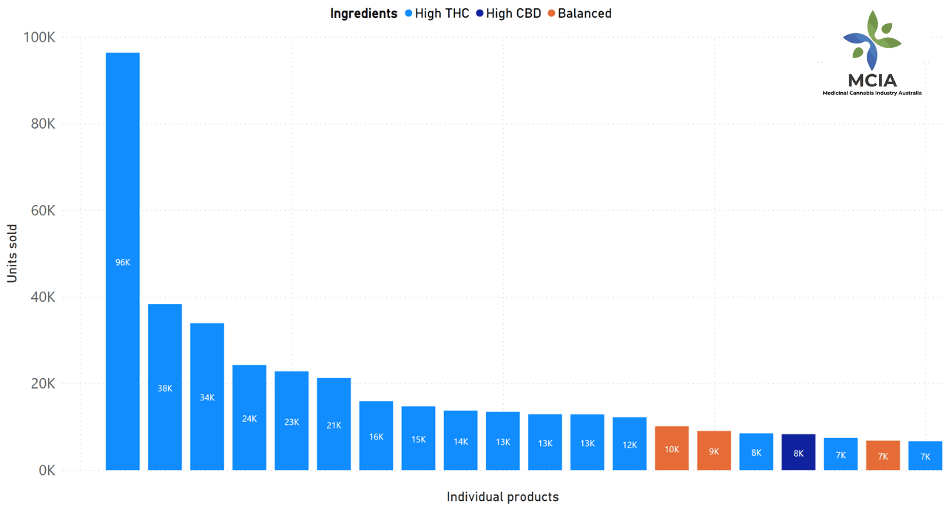
The market is dominated by a small number of popular products. Although sales were reported for 360 different products, the top 10 products account for 42.3% of total units sold (294,680 units). [FIGURE 2] The most popular individual product sold (96,265 units) represented less than 14% of all units sold. Mr Crock said “The top 10 products were all dry herb and inhalable products, supporting the observed trend of strong growth in flower products”. Of the top 20 products, five were oral oils, and 15 were dry herb and inhalable products.
By category, of the top 20 products, one was high-CBD, three were balanced, and 16 were high-THC products. [FIGURE 3]
This new data is important for the industry to better understand patient and prescriber needs. MCIA Chair, Mr Peter Crock, said “It is critical that the Government provides access to industry wide data to assist the industry to effectively plan and respond to patient needs.”
Download the PDF or DOC version of this Media Release
MEDIA CONTACTS
Peter Crock
Chair
0419 307 467
Rosemary Richards
Executive Manager
0411 564 128
About the data:
Companies that have legal ownership and responsibility for medicinal cannabis products in Australia (product “sponsors) are required by the Commonwealth government to report the number of units sold every 6 months. This data was provided by the Therapeutic Goods Administration (TGA) within the Department of Health to Medicinal Cannabis Industry Australia via a Freedom of Information request.
While product sponsors are required to report these figures completely and accurately, the TGA cannot guarantee that all sponsors have met their legal obligations, and the TGA assumes no legal liability or responsibility for the accuracy, currency, completeness, or interpretation of the information.
Data from sponsors does not include compounded medicinal cannabis products.
About MCIA:
MCIA is the peak industry organization and voice for Australia’s medicinal cannabis industry. Our focus is on building a legitimate industry that enhances wellbeing through facilitating access to quality Australian medicinal cannabis products for Australian and global patients. MCIA provides stewardship for an economically sustainable and socially responsible industry that is trusted and valued by patients, the medical community and governments. The Australian industry and its products are built on sound science and underpinned by industry processes and standards that ensure patients, the medical community and governments have confidence in the sector and its products.
Figure 1: Units sold by dosage form
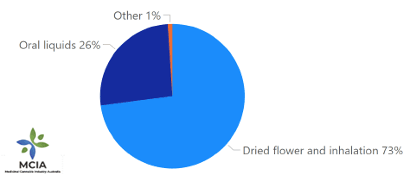
Figure 2: Top products sold by unit (Dosage form)
Light blue: Fried flower and inhalation
Dark blue: Oral liquids
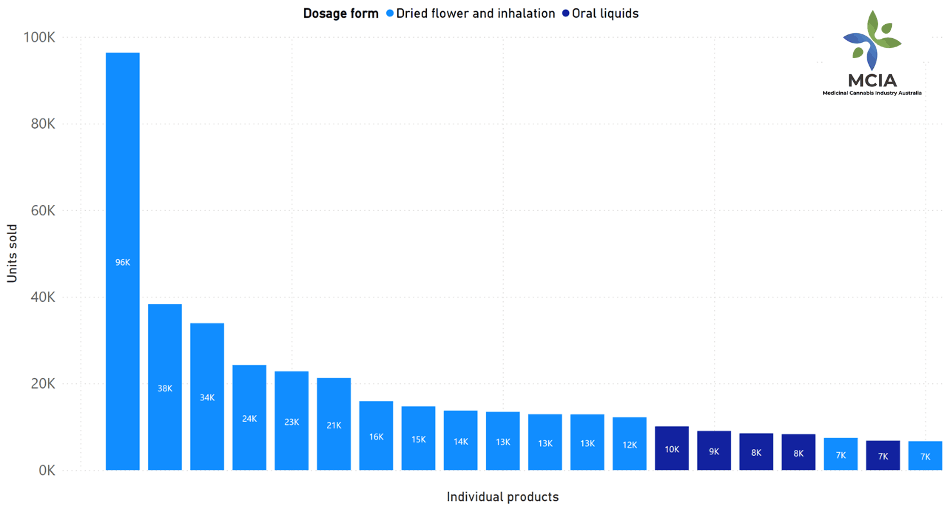
Figure 3: Top products sold by unit (ingredients)
Light blue: High THC
Dark blue: High CBD
Orange: Balanced